Product disclosure for products in Vietnam ensures that goods meet national safety and quality standards before entering the market. Importers and local manufacturers submit specific technical documents to government authorities. Regulators review the product dossier to confirm compliance with Vietnamese commercial laws. The Ministry of Health (MOH) and the Vietnam Food Administration (VFA) manage different product categories. This guide explains the regulatory authorities, required documents, and submission steps for successful product compliance in the Vietnamese market.
What does product disclosure mean in Vietnam?
Product disclosure in Vietnam means registering or declaring a product with the authorities before sale or distribution. The importer or manufacturer submits a dossier to prove safety, quality, labeling, and regulatory compliance. The Vietnamese government requires this legal process to protect public health and consumer rights. The procedure applies equally to imported goods and locally manufactured items. Regulators evaluate the ingredients, usage instructions, and foreign safety certificates. Successful disclosure results in a unique registration number or a notification receipt. Businesses print this disclosure number on the local product label or keep the digital receipt on file for market inspections.
Key entities involved in this process:
- Vietnam Ministry of Health (MOH)
- Vietnam Food Administration (VFA)
- Department of Medical Equipment and Construction
- Importers
- Manufacturers
Why do companies need product disclosure in Vietnam?
Companies need product disclosure in Vietnam to legally import, manufacture, and sell regulated goods in the local market. Customs officials require approved disclosure documents to clear commercial shipments. Market surveillance teams frequently check retail stores and warehouses for regulatory compliance. Selling goods without a valid disclosure receipt causes severe legal trouble and financial penalties. The disclosure process builds consumer trust by proving the physical product meets national health standards. Distributors and supermarket chains actively require the disclosure number before stocking new items on their shelves.
What is the difference between disclosure and registration?
Product disclosure applies to lower-risk goods via notification, while product registration applies to high-risk goods requiring intense clinical evaluation. Both processes result in a market authorization number. Cosmetics and standard packaged foods typically undergo a disclosure or notification process. The authority receives the dossier and issues a receipt based on the applicant's declaration. High-risk items, such as pharmaceuticals and medical devices, require formal registration. Registration involves rigorous laboratory testing, clinical trial reviews, and longer government approval timelines.
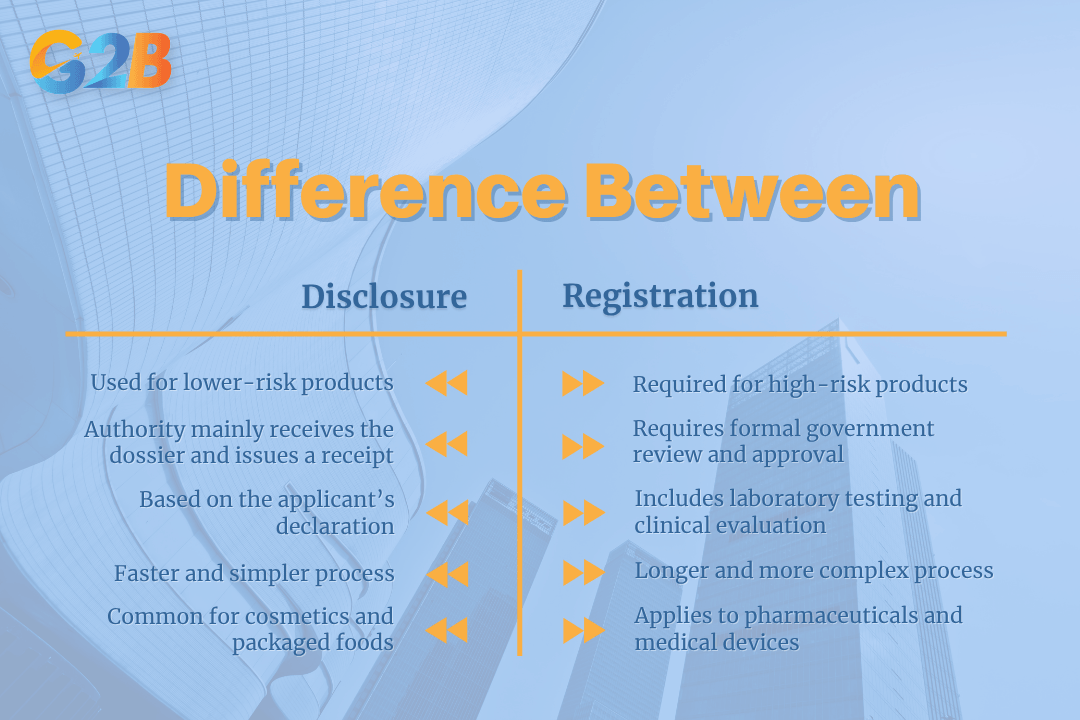
There are many differences between disclosure and registration
Which authorities regulate product disclosure in Vietnam?
Different Vietnamese authorities regulate disclosure depending on the product category. The Ministry of Health, the Vietnam Food Administration, and the Ministry of Industry and Trade handle distinct types of goods. The regulatory framework dictates which government office receives the application dossier. Submitting documents to the wrong authority causes immediate application rejection.
| Product category | Regulatory authority | Legal framework |
|---|---|---|
| Food & supplements | Vietnam Food Administration (VFA) | Law No. 17/2018/QH14 on Food Safety |
| Cosmetics | Drug Administration of Vietnam (DAV) | Circular 06/2011/TT-BYT implementing ASEAN Cosmetic Directive |
| Medical devices | Ministry of Health (MOH) | Decree 98/2021/ND-CP, amended by Decree 07/2023/ND-CP |
| Chemicals | Ministry of Industry and Trade | Law No. 06/2020/QH14 on Chemicals |
Key roles of regulatory authorities:
- Review compliance documents and ingredient lists
- Issue disclosure numbers or confirmation receipts
- Monitor post-market compliance and product safety
What is the role of the Vietnam Food Administration (VFA)?
The Vietnam Food Administration (VFA) regulates food safety, dietary supplements, and food additives. The VFA reviews product dossiers to ensure raw ingredients meet national public health standards. The VFA operates directly under the Ministry of Health. Companies submit food disclosure applications to the VFA via a centralized online portal. The agency checks the Certificate of Analysis (COA) and assesses the proposed product labels. The VFA issues a confirmation of food safety compliance for successful applications.
What is the role of the Drug Administration of Vietnam (DAV)?
The Drug Administration of Vietnam (DAV) manages the disclosure and registration of cosmetics and pharmaceutical products. The DAV ensures that products comply strictly with the ASEAN Cosmetic Directive. Importers submit cosmetic notification dossiers to the DAV electronically. The agency verifies the submitted ingredient list against international banned or restricted substance lists. The DAV provides an official cosmetic notification receipt number. Businesses use this exact receipt to clear cosmetic shipments through Vietnam Customs.
Which products require product disclosure in Vietnam?
Vietnam requires product disclosure for regulated goods before import, manufacturing, or commercial distribution. Common regulated items include health supplements, cosmetics, packaged foods, and medical devices. The government classifies physical products based on their potential impact on human health and safety. Unregulated products, like basic office stationery, do not need this formal disclosure process. Items applied directly to the human body or consumed internally face strict regulatory rules.
Common product categories requiring disclosure:
- Declare dietary supplements
- Register functional foods
- Notify cosmetics
- Declare packaged foods
- Register food additives
- Notify medical devices
Do cosmetic products require disclosure in Vietnam?
Yes, all cosmetic products require a cosmetic product notification in Vietnam before entering the retail market. The importer must submit the formula and packaging details to the Drug Administration of Vietnam. Cosmetics include skincare lotions, makeup, haircare serums, and perfumes. The product formula must follow the guidelines of the ASEAN Cosmetic Directive. The foreign manufacturer provides a comprehensive list of ingredients with accurate percentage weight levels. The DAV checks the formulation to detect any prohibited chemicals.
Do dietary supplements need disclosure in Vietnam?
Yes, dietary supplements need a formal product declaration with the Vietnam Food Administration. Supplements face stricter scientific scrutiny than regular packaged foods due to their specific health claims. The submitted dossier for health supplements must include clinical evidence or scientific research documents supporting the advertised health claims. The foreign manufacturer must hold a valid Good Manufacturing Practice (GMP) certificate. The VFA reviews the recommended daily dosage, active ingredients, and mandatory warning statements.
Do medical devices require notification in Vietnam?
Yes, medical devices require classification and notification with the Ministry of Health. Class A and Class B devices undergo a standard declaration, while Class C and Class D require formal registration. Medical devices include items ranging from simple surgical masks to complex MRI machines. The applicant must classify the device based on its inherent risk level. The dossier requires quality management certificates, such as ISO 13485, and clear technical manuals.
What documents are required for product disclosure in Vietnam?
Vietnamese authorities require a product dossier proving safety, origin, and regulatory compliance. The local importer collects certificates from the foreign manufacturer to build the compliance file. The exact documents change based on the specific product category. Core documentation requirements remain highly similar across all government departments. Authorities demand original documents or legally certified copies of foreign certificates.
A typical disclosure dossier includes:
- Submit the product declaration form
- Provide the Certificate of Free Sale (CFS)
- Include the Certificate of Analysis (COA)
- Attach the manufacturing license or GMP certificate
- Present the product label and packaging artwork
- List all product ingredients
- Supply the technical product specification sheet
Additional documents for imported products:
- Provide a Power of Attorney from the manufacturer
- Submit notarized and legalized certificates
- Include Vietnamese translations of technical documents

Required documents for product disclosure in Vietnam
What is a Certificate of Free Sale (CFS)?
A Certificate of Free Sale (CFS) is a government-issued document proving that a product has been legally sold in the exporting country. Vietnamese authorities require the CFS for imported cosmetics, supplements, and medical devices. The national health or commerce authority in the manufacturer's home country issues the CFS. The CFS must state the exact product name and the manufacturer's registered address. Vietnam requires the CFS to have proper consular legalization. The document proves the product successfully meets the safety standards of its home market.
What are the requirements for a Certificate of Analysis (COA)?
A Certificate of Analysis (COA) must show the physical, chemical, and microbiological testing results of a product batch. An independent ISO 17025-accredited laboratory usually issues the COA. The COA verifies that the manufactured product matches its technical specification sheet. The VFA requires a COA issued within 12 months before the formal application date. The document must list the testing methods, acceptable reference limits, and the actual test results for heavy metals and microbes.
What is the product disclosure process in Vietnam?
The disclosure process involves preparing documents, submitting the dossier to the authority, and receiving approval or a receipt number. The applicant must use the designated government online portal. The procedure requires exact attention to technical details. A minor typo in the paperwork causes immediate application rejection. Companies follow a strict linear process to secure their product notification number.
Typical disclosure process:
- Prepare documentation: The importer gathers certificates, product specifications, and original labels from the foreign manufacturer.
- Legalize foreign documents: Authorities require consular legalization for foreign certificates at the Vietnamese Embassy.
- Submit the dossier: The importer submits the files through the Vietnam public service portal or regulatory office.
- Authority review: Government officials review commercial compliance, ingredient limits, and labeling accuracy.
- Receive disclosure confirmation: The authority issues a product declaration number or notification receipt.
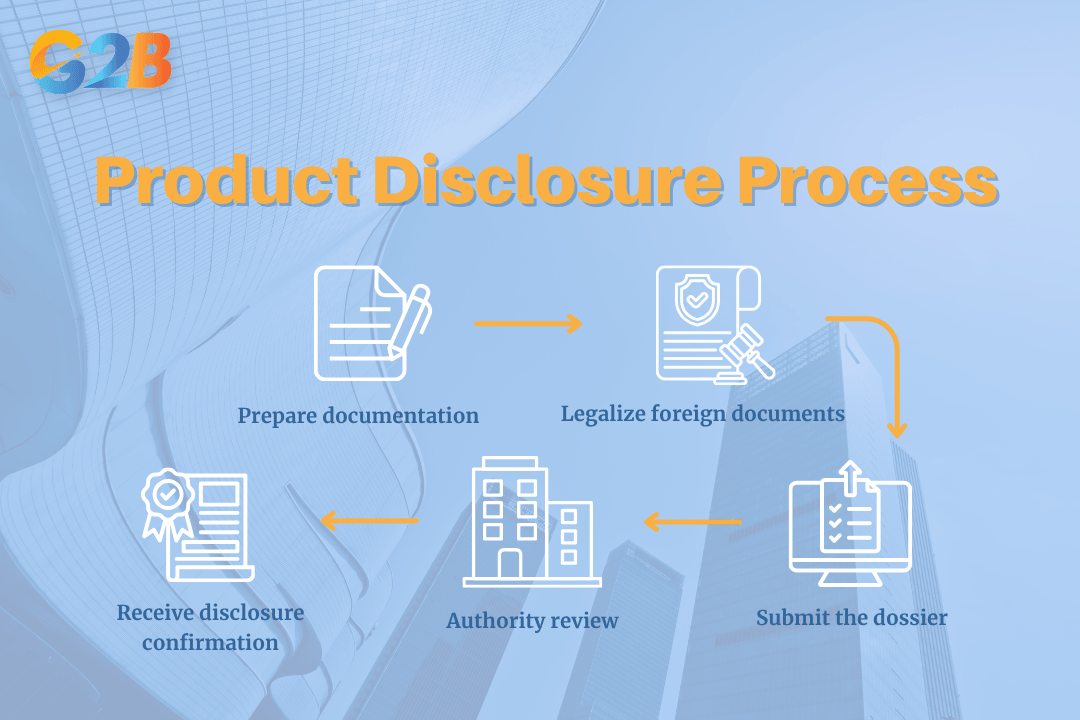
Five steps of the product disclosure process in Vietnam
How to legalize foreign documents for Vietnam?
Foreign documents need consular legalization to be legally valid inside Vietnam. The exporter notarizes the document, certifies it at their local foreign affairs department, and stamps it at the Vietnamese Embassy. Vietnam does not accept the Apostille stamp for many international trade documents. The multi-step legalization process proves that the signature and official seal on the foreign document are genuine. Documents like the CFS, Letter of Authorization (LOA), and GMP certificate absolutely require this step. Consular legalization takes several weeks, requiring companies to plan their supply chain timelines carefully.
How do authorities review the application?
Authorities review the application by comparing the submitted documents against national technical regulations. Officials verify the ingredient safety limits, check document validity, and assess the proposed Vietnamese label. If the digital dossier is correct, the authority issues the final approval receipt. If the dossier contains regulatory errors, the authority issues an official letter requesting specific amendments. The applicant then has a strictly limited time frame to correct the mistakes and resubmit the digital files.
How long does product disclosure take in Vietnam?
Product disclosure processing time depends on the product category and dossier completeness. Routine cosmetic notifications take a few days, while health supplements require several weeks of evaluation. The statutory timelines often differ from the practical working timelines. Government departments handle high volumes of trade applications. Significant delays occur if the applicant submits incorrect data or uncertified documents.
| Product type | Typical processing time |
|---|---|
| Cosmetics | 3 - 7 working days |
| Packaged food | 7 - 21 working days |
| Health supplements | 20 - 30 working days |
| Medical devices | 10 - 30 working days |
Factors affecting approval timelines:
- Submit missing documents
- Provide incorrect ingredient classification
- Design label non-compliance
How long is the disclosure receipt valid?
The validity of a product disclosure receipt depends entirely on the product type. Cosmetic notification receipts remain valid for five years, while standard food declarations often possess an unlimited validity period. Importers must renew cosmetic receipts before they officially expire to continue selling the goods legally. If the foreign manufacturer changes the core product formula, the importing company must apply for a brand-new disclosure number. Minor administrative changes, like an updated importer corporate address, merely require a supplementary notification.
What are Vietnam's labelling requirements for disclosed products?
Vietnam requires product labels in the Vietnamese language before retail sale. The printed label must accurately reflect the exact information approved in the product disclosure dossier. Government Decree 43/2017/ND-CP governs commercial goods labeling in Vietnam. The original foreign language label can remain on imported goods, but a supplementary Vietnamese text decal is legally mandatory. The label must clearly inform the local consumer about the product's origin, intended usage, and health warnings.
Mandatory label information:
- Print the product name
- List the ingredients
- Display the manufacturer's name and address
- State the country of origin
- Show the expiry date
- Include the batch number
- Provide usage instructions
- Add the importer information
How to apply supplementary labels for imported goods?
Importers apply supplementary Vietnamese labels over the original packaging before releasing the goods to the local market. The supplementary label must never obscure essential safety information on the original package. Customs officers inspect commercial shipments at the seaport to ensure labeling compliance. Importers often stick the translated labels onto the products at a licensed bonded warehouse before final customs clearance. The Vietnamese text on the supplementary label must precisely match the approved product declaration dossier. Discrepancies between the physical label and the digital dossier trigger customs clearance rejections.
What happens if a product is sold without disclosure in Vietnam?
Vietnamese regulators impose strict administrative penalties for selling undisclosed regulated products. Market surveillance authorities continuously inspect import shipments, retail stores, and online e-commerce sellers. The central government actively monitors the consumer market to protect citizens from unsafe or counterfeit goods. Selling unregistered cosmetics, foods, or medical devices severely violates local commercial laws. Regulators perform random physical checks on warehouse inventories and retail supermarket shelves.
Possible penalties include:
- Execute product seizure
- Issue fines between 20 million and 100 million VND
- Order mandatory product recall
- Enforce business license suspension
How do authorities monitor post-market compliance?
Authorities monitor post-market compliance by purchasing random product samples from the market and testing them in government laboratories. Inspectors check if the physical product matches the previously declared dossier. The local Department of Health conducts periodic, unannounced inspections on authorized local distributors. If a tested product contains dangerous undeclared ingredients, the health authority immediately issues a national recall notice. The government publicly publishes the names of violating companies and dangerous products on official regulatory websites.
What are common product disclosure mistakes in Vietnam?
Companies frequently delay approvals by submitting incomplete or non-compliant dossiers. Minor errors in the technical paperwork cause regulatory authorities to reject the entire application. Preparing a flawless disclosure application requires a deep technical understanding of Vietnamese regulations. Foreign manufacturers frequently provide internal documents that fail to meet Vietnam's highly specific formatting demands. Local importers often fail to cross-check the foreign documents carefully before portal submission.
Common mistakes to avoid:
- Submit incorrect ingredient classification
- Omit the missing Certificate of Free Sale
- Upload non-legalized documents
- Fail to translate labels into Vietnamese
- Provide inconsistent product specifications
How does incorrect ingredient classification affect the application?
Incorrect ingredient classification causes the regulatory authority to automatically reject the disclosure application. Products containing borderline medicinal substances face forced reclassification from cosmetics or supplements directly into pharmaceuticals. Vietnam maintains heavily regulated lists of permitted food additives and safe cosmetic ingredients. If an importer declares a cosmetic product that secretly contains a banned substance under the ASEAN Cosmetic Directive, the application instantly fails. Companies must carefully cross-check their exact ingredient formulas with the latest Vietnamese regulatory annexes before applying.
Who can submit a product disclosure in Vietnam?
Only a Vietnam-registered company can submit a formal product disclosure application. The applying local entity acts as the legally responsible party for the product's quality and safety in the domestic market. Foreign manufacturers cannot submit the electronic dossier directly from overseas. They require a local corporate representative to handle the ongoing legal responsibility. The local company uses its secure digital signature token to upload PDF documents to the national government portal. The local company name permanently appears on the official disclosure receipt and the final printed product label.
Eligible applicants include:
- Act as Vietnamese importers
- Operate as local manufacturers
- Serve as authorized distributors
- Work as regulatory consulting firms with authorization
Can foreign companies submit product disclosure directly?
Foreign companies cannot submit product disclosure directly in Vietnam. Foreign companies must formally appoint a Vietnamese legal entity as the responsible applicant to manage the regulatory submission. The foreign manufacturer officially signs a Letter of Authorization (LOA) assigning a local distributor or local subsidiary as the exclusive applicant. The LOA must clearly state the specific product names and the exact scope of the legal authorization. This document ensures the Vietnamese government possesses a local corporate entity to hold financially accountable for product recalls or public safety issues.
Product disclosure for products in Vietnam remains an absolute legal requirement for successful market entry. Importers and local manufacturers must fully understand the technical rules set by the Ministry of Health, the Vietnam Food Administration, and other regulatory bodies. Preparing a highly accurate application dossier with legally certified documents, correct ingredient classifications, and translated labels prevents costly supply chain delays. A proactive, detail-oriented approach to regulatory compliance protects the international brand reputation and ensures profitable, long-term business success inside the Vietnamese market.
Planning to launch your business in Vietnam? Get in touch with G2B for expert guidance on setting up a company in Vietnam and ongoing annual compliance. We will take care of the regulatory procedures, so you can concentrate on growing your business.


 Delaware (USA)
Delaware (USA)  Vietnam
Vietnam  Singapore
Singapore  Hong Kong
Hong Kong  United Kingdom
United Kingdom 
